News Story
Ingestible device research advances, enters new phase

[Click photo for larger image] Schematic with dimensions (top) and scanning electron microscope (SEM) images (bottom) of a barbed microneedle fabricated via 3-D DLW. The zoomed-in image depicts the high-fidelity fabrication of the curved barbs with sharp tips (~ 1 μm resolution). Figure 2 from the IEEE MEMS 2020 paper, Biomimetic Barbed Microneedles for Highly Robust Tissue Anchoring.
Recently there have been many advances in the development of minimally invasive medical devices, paving the way for ingestible capsule systems to detect and subsequently treat/monitor chronic gastrointestinal (GI)-associated pathologies, including inflammatory bowel diseases like Crohn's disease and ulcerative colitis. Such devices can now perform GI imaging, gas sensing, lesion biopsy, and drug release. However, a number of practical problems remain to be solved before the promise of these devices can become a reality.
Professor Reza Ghodssi (ECE/ISR), his faculty colleagues and his research group in the MEMS Sensors and Actuators Laboratory are continuing to make important advances in ingestible device research. Now, new funding from the National Science Foundation and a paper just published in the journal Lab on a Chip are further extending their research.
New funding
Ghodssi is the principal investigator for a new three-year, $350,000 award from the National Science Foundation (NSF), Closed-Loop Sensing and Actuation for Gastrointestinal Capsule Systems.
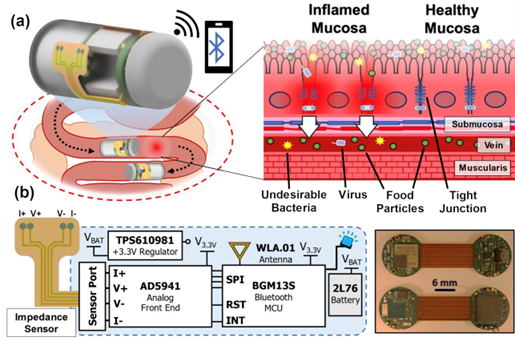
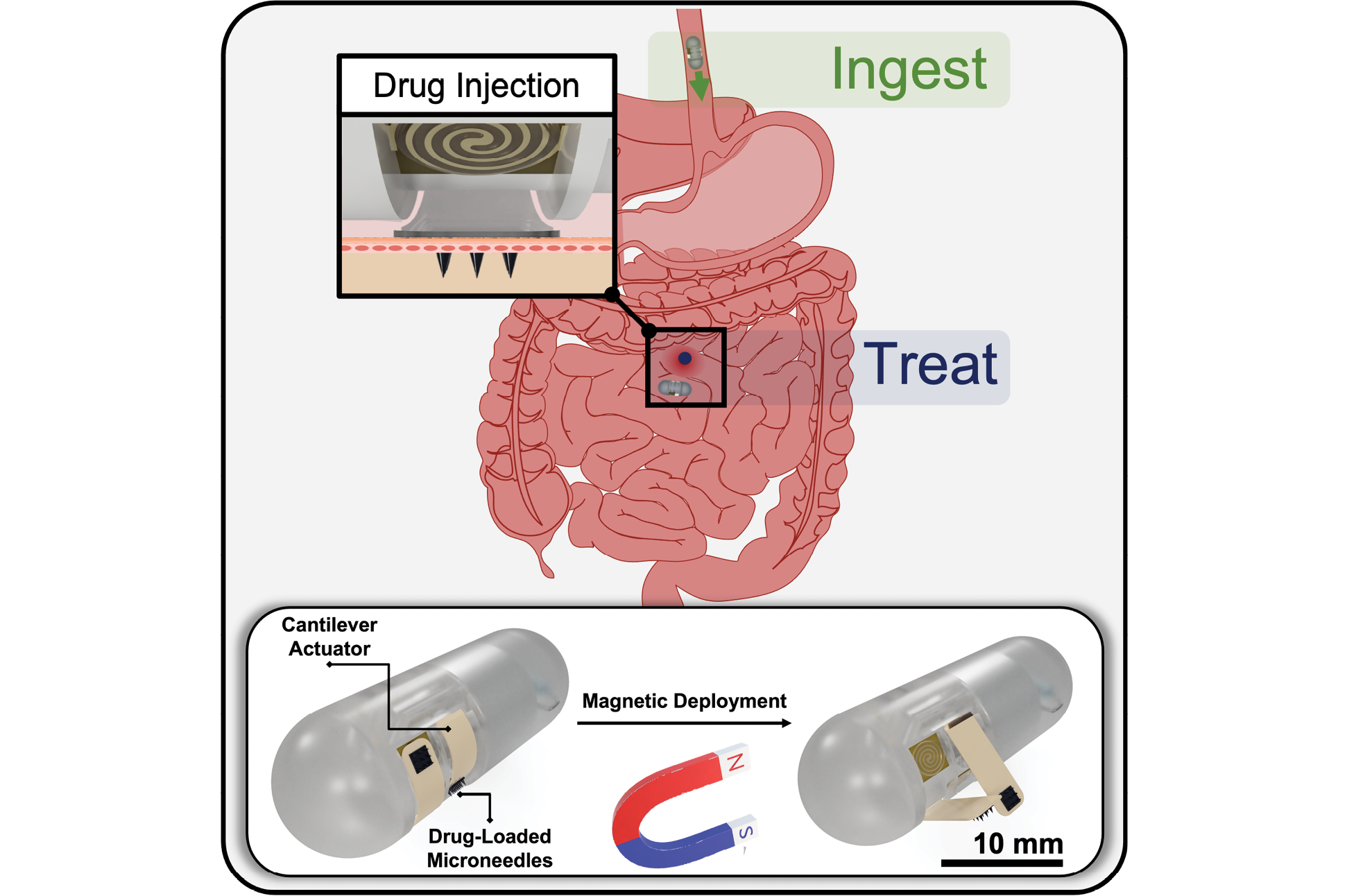
The grant will enable Ghodssi and his research group to develop an ingestible capsule featuring a closed-loop operation capable of sensing inflammation in the GI tract and treating it with medication-laden “microdarts.” A sensor signal will trigger the capsule to release and anchor the microdarts into the mucus that lines the GI. This integrated capsule system will enable effective detection, intervention, and further surveillance of GI pathologies.
Few systems have demonstrated feedback-driven intervention in response to sensor signals. This is because of the challenges inherent in developing robust sensor technologies that can operate in the GI environment and the requirements for compact actuators to apply interventions. The small form factor and power requirements for ingestible systems have amplified these challenges.
In the new research, a feedback-driven system integration will provide a technological platform with three innovations: microdart-loaded thermomechanical spring actuators for targeted GI diagnostics and treatment, systems integration of sensor-enabled GI tract-targeting capsules, and in vitro model design and system validation. The delivery, actuation, and anchoring mechanisms of the capsule systems will be tested in simulated benchtop models, including one made from synthetic tissue that can mimic peristalsis of the GI tract and another simple model that uses animal intestines with tubing to inject solutions for simulating GI secretions.
Early work on microdarts was presented in S. Liu, S. Chu, G. E. Banis, L. A. Beardslee, and R. Ghodssi, “Biomimetic Barbed Microneedles for Highly Robust Tissue Anchoring,” at the 33rd IEEE International Conference on Micro Electro Mechanical Systems (IEEE MEMS 2020), pp. 885-888, Vancouver, January 2020.
New Lab on a Chip paper
Alumni George Banis (BioE Ph.D. 2019) and Ragendramayavan Sathyam (Robotics M.Eng. 2019); UMD Research Associate Luke Beardslee, M.D., Ph.D.; ECE Ph.D. student Justin Stine; and Professor Reza Ghodssi (ECE/ISR) have written Capacitive sensing of triglyceride film reactions: a proof-of-concept demonstration for sensing in simulated duodenal contents with gastrointestinal targeting capsule system, published in the Royal Society of Chemistry’s April 2020 Lab on a Chip.
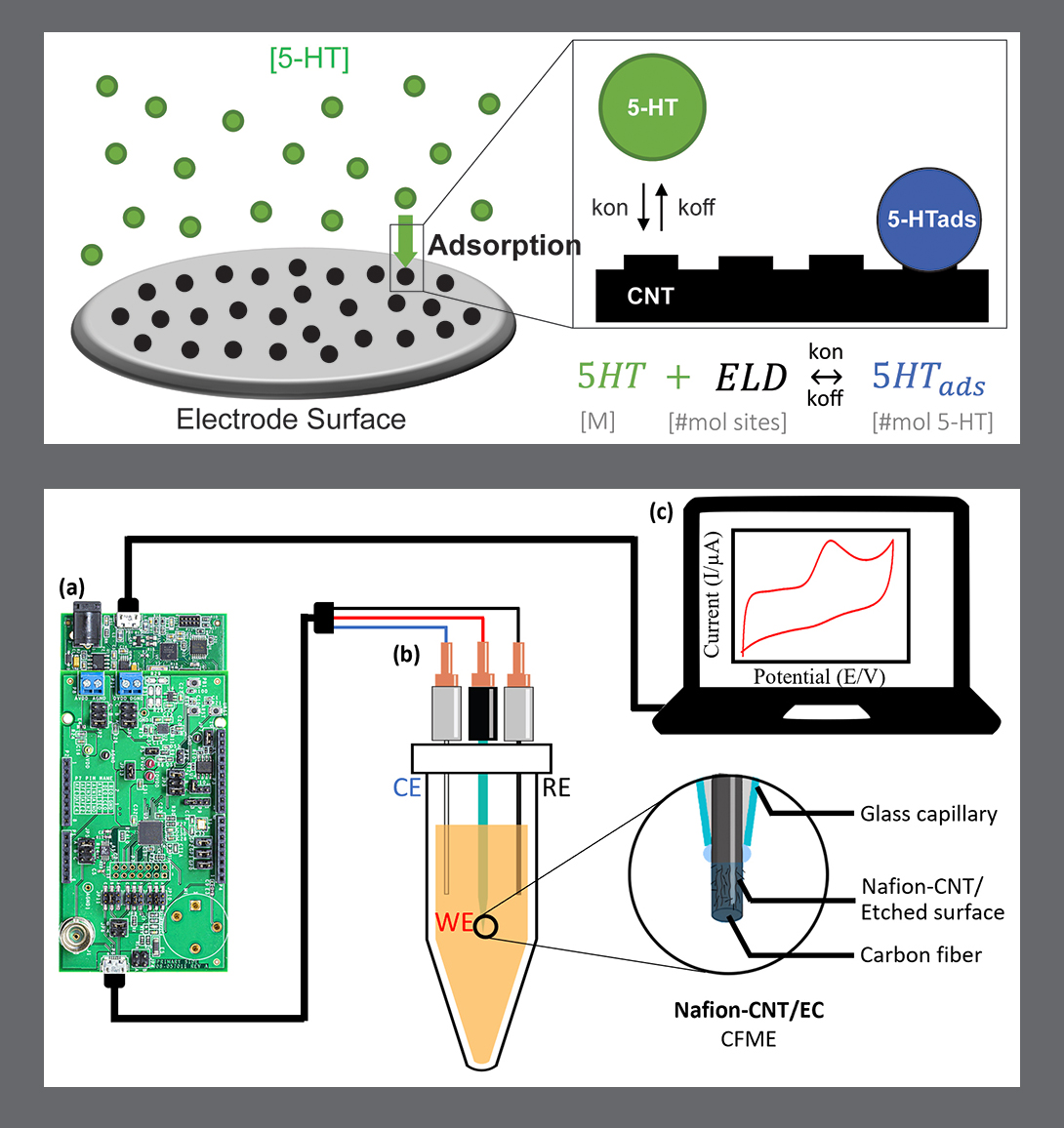
The paper describes an integrated ingestible capsule sensing system with triglyceride film-coated capacitive sensors that can measure biochemical species such as pancreatic lipase and bile acids in the duodenum. The microfabricated capacitive sensors communicate via a Bluetooth low-energy (BLE)-microcontroller, which allows wireless connectivity to a mobile app.
The capsule’s 3D-printed package is coated with polymers that remain intact in an acidic solution (similar to gastric conditions), and dissolve at a duodenum-mimicking neutral pH. This triggers an opening of the sensing chamber from which the presence of pancreatic lipase can be detected—a significant step towards using embedded packaging and triglyceride-based materials to target specific regions of the GI tract and sense biochemical contents for evaluating gastrointestinal health.
Building on current and past research
The new funding and research results build on the group’s successes in ingestible devices.
Ghodssi, William Bentley (BioE/Fischell Institute/IBBR), Associate Professor Jens Herberholz (Psychology/NACS), and Professor Wolfgang Losert (Physics/IPST/IREAP) are currently in the midst of a $1M NSF project, “Developing engineering solutions to investigate microbiome-to-neuron communication.” This research is providing a more realistic picture of the complex gut-microbiome-brain axis system (GMBA). The simple, novel and customizable platform being developed will provide a fuller picture of the GMBA. Pilot data for the project was gathered through a seed grant award from UMD’s Brain and Behavior Initiative (BBI) for “A Multimodal Sensor Discovery Platform to Study the Molecular Events Underlying the Gut-Microbiome-Brain-Axis.”
In addition, this March Ghodssi’s group published an ingestible device research review paper in the American Chemical Society’s journal ACS Sensors.
“Ingestible Sensors and Sensing Systems for Minimally Invasive Diagnosis and Monitoring: The Next Frontier in Minimally Invasive Screening" covers the development of and uses for ingestible electronic sensing systems, particularly within the GI tract. It examines conventional techniques, as well as ingestible sensors and sensing systems that are currently under development for use in disease screening and diagnosis for GI disorders, including their design, fabrication and applications.
It was written by UMD Research Affiliate Luke Beardslee, M.D., Ph.D.; alumnus George E. Banis (BIOE Ph.D. 2019); former postdoctoral researchers Sanwei Liu and Sangwook Chu (also a EE Ph.D. 2018 alumnus); Bioengineering Ph.D. student Ashley Chapin; ECE Ph.D. student Justin Stine; Pankaj Jay Pasricha, a Johns Hopkins University Professor of Medicine and Director of the Johns Hopkins Center for Neurogastroenterology; and Ghodssi.
Ghodssi believes in the power of academic research groups writing review papers to summarize a field they have come to know well as they work together on a major project. “It allows us to expand our view to a field so we are not just focused on one specific work,” he says.
Published May 26, 2020